ABDO have made us “Charity of the Month”. You can read their article here
Blog Posts
Sight Village
Queen Alexandra Colledge is delighted to be returning to Eastside Rooms Conference Centre, Birmingham, for a two day event on Monday 7th and Tuesday 8th July 2025. The event is free to visit. There will be a wide range of exhibitors in attendance. We would recommend that you pre-register your attendance by clicking this link
This is a great opportunity to see the latest technology for people with sight problems and see what other help is available. Visit the Sight Village Website.
The full address of the venue is:
Eastside Rooms Conference Centre, Woodcock Street, Birmingham, B7 4BL
Monday – Doors open at 10.00am until 4.00pm
Tuesday – Doors open at 9.30am until 3.30pm.
Do stop by and say hello or help out manning the stall and chatting to visitors interested in our condition.
Research update 3 shows promising results
If you have been following our previous posts; Engineers to develop spectacle lenses for keratoconus patients, Research update and Spectacles for KC Research update 2, you will be aware of Dr Ahmed’s work to address the problem of non-orthogonal astigmatism.
At our AGM and Speaker meeting both Dr Abass and optometrist Lynn White, carefully explained the progress of their work to find a solution for those of us who have to cope with not wearing contact lenses for a period of time; yet still need better vision for dealing with basic tasks. Both Ahmed and Lynn are convinced that with the advance of technology, spectacles can be manufactured to take account of a non-orthogonal angle of astigmatism. Indeed, some who have worn the spectacles in laboratory conditions, do not want to give them back.
In this clip, Dr Abass explained how he was able to interpret the high-resolution results without relying on the software provided by modern topography machines. He noticed that astigmatism is not always orthogonal, which led to his research project to find a better solution for keratoconus.
Here Lynn White explains the wider implications of Dr Abass’ research beyond keratoconus.
There is still a long way to go but results to date are promising. More details will be given in our next newsletter.
Here is the press release from Liverpool University.
In a recent KC patient group meeting, Dr Ahmed Abass from the University of Liverpool unveiled exciting research into a new type of spectacle lens designed to help those living with keratoconus; a progressive eye condition that causes thinning and irregular shaping of the cornea.
Traditional glasses often fall short in correcting vision for keratoconus patients due to a phenomenon called non-orthogonal astigmatism. However, Dr Abass and his team, supported by Fight for Sight and the UK Keratoconus Self-Help and Support Association, have been exploring an innovative lens design that targets this issue directly.
The results are encouraging. In this pilot study, many participants experienced clearer vision, reduced ghosting, and sharper letter recognition with these specially designed lenses.
“This is the first study of its kind,” Dr Abass said. “We’ve shown that the idea works. Now we need to focus on scaling it up, finding ways to mass produce these lenses and test them on a larger group.”
As the project looks towards the next stage, development and commercialisation, there may be exciting opportunities for those in the optical or medical device industries to get involved. Anyone interested in collaborating with the University of Liverpool or learning more is welcome to contact Dr Abass at a.abass@liverpool.ac.uk. or connect via linkedin
It is a small but meaningful step forward, offering new possibilities for those seeking alternatives to contact lenses or invasive procedures.
https://www.liverpool.ac.uk/people/ahmed-abass
New Research Study
Investigator Initiated, Prospective Study of Xenia Corneal Lenticule
This information is provided directly by researchers and we recognise that it isn’t always easy to understand. We are working with researchers to improve the accessibility of this information.
Cornea ectatic conditions such as keratoconus and post-LASIK (Laser In situ Keratomileusis) ectasia are disorders of the eye that are notorious for its uncontrolled progression over time, leading to loss of vision. There are valuable treatment options for controlling disease progression in mild to moderate stages of such disease. However in advanced cases and / or when contact lenses cannot be worn, surgery remains the only treatment option to rehabilitate vision. Most treatment options are highly invasive and represent significant risks. This study evaluates a novel corneal implant, Gebauer™ Lenticule to treat severe keratoconus or post-LASIK ectasia. This implant is derived from porcine collagen and intended for intra-stromal insertion. The Gebauer™ Lenticule is expected to improve the stability of the cornea while not impairing the vision. The procedure is an additive procedure after other treatment options have been exhausted. The procedure is reversible with removal of the implant (in the unexpected case of an adverse reaction), and vision may be restored to baseline.
The purpose of this study is to evaluate how well the implant is tolerated, it’s safety profile, and how effective this new treatment option is in the treatment of keratoconus or post-LASIK ectasia.
Visit the website for more details – here
Keratoconus in my teens/twenties
Around the age of 15/16 I was diagnosed with keratoconus. I was a glasses wearer, but I was really struggling to see anything in much detail. Trying to see the board at school was tough, even close up reading became a challenge. I spent most of my GCSE years squinting, much to my embarrassment. None of my friends had the same difficulties that I had. My Mom requested large print for my exams, and although at the time I felt pretty mortified by it, in hindsight the larger font and extra time was a big help – thank you Mom…..
That Summer, I was given rigid gas permeable lenses to try. I’ve always been very sensitive about anyone going near my eyes (even now at the age of 30, during a regular check up at the eye hospital it can be a challenge!), so someone putting these uncomfortable, foreign objects in my eyes was quite the ordeal. Especially considering the way I was taught to remove them was with a mini plunger – terrifying! Regardless, due to the steep improvement in vision with these RGP lenses, I persevered, and eventually got used to them. I even got used to wearing the single lens if one of my eyes was giving me some grief. My vision had improved and my confidence was up.
However, it wasn’t all good times in the coming years. I learnt the hard way that rubbing your eyes can make your vision worse (I haven’t done that now for 13 years and counting…..), I was given new RGP lenses which would prioritise comfort rather than sight, even having to change to piggyback lenses in one of my eyes as it couldn’t cope with just the RGP anymore, and I was told on a few occasions that my condition was worsening of its own accord. The lenses themselves could be quite uncomfortable at times, and the slightest speck of dust could often send me into full on tears! And all this time being a young adult trying to navigate A-Levels and then university, I didn’t really listen to any advice I was being given, and I just got frustrated and angry at my hospital appointments. I wonder if the feeling may have been mutual from the hospital however, as I was constantly asking for replacement contact lenses due to them flicking out. Unfortunately when you’re experiencing the uni lifestyle of bars and nightclubs, the dehydration from a night out means that all I needed is a stray hand in and around my eye to make my contact lens flick out. There goes another payment (sorry Mom!) and many weeks waiting for the new lens to arrive…..
Fast forward to adult life in a full time employment at the age of 25 ,and I passed my driving test. I was always nervous about driving – I didn’t always have confidence in my sight (regardless of whether I could read the required line on the eye chart or not) and thought that it might affect my driving ability. After a few years of having lessons on and off, I finally booked a test. Weirdly the thing I was most nervous about on the day wasn’t actually the driving, but reading the reg plate in the car park!
I feel that this is the point where my outlook changed. I could drive, and once I got my first car, I suddenly had way more opportunities in my life that were not there previously, in particular for work and travel. Now that I had this freedom, I really did not want to lose it. I started to take more care in my day-to-day life – less wear time with the contact lenses where possible, more time and care with cleaning and storing the lenses, I even started to wear Edgar Davids-esque protective goggles for sport. I didn’t care if I got the odd comment – I needed good eye health now more than ever.
During a regular eye check-up at the hospital, I was told once again that my condition had deteriorated, and that once again, I should undergo cross linking to stop the condition from getting any worse. This operation is something that I had been told about on a few occasions previously. However, with my lack of affection of anyone or anything going near my eyes, as well as turning a bit of a blind eye to my condition in years gone by, I had always ignored the suggestion. This time though, I felt like I had something that I wanted to preserve – my eyesight for driving. I reluctantly agreed to be put forward for cross linking (under the condition of being under general anaesthetic, you can touch my eyes but I don’t want to be awake to see it!), and to my surprise within two months I was in hospital having the operation. I have written a piece previously about this operation, link as follows:
Before my operation, I was very nervous and I felt like I needed reassurance from somewhere. For many years, my Mom had been in contact with the West Midlands Keratoconus Group, even going to a few of the meet ups in Birmingham. Although her previous suggestions for me to join had fallen on deaf ears, this time I decided to join the latest Zoom call. It was an extremely eye-opening experience. Not only was I not alone with some of my experiences, they were in fact commonplace. Finally I had met some other people who were also fed up of the question ‘why can’t you wear glasses?’ Some of the members really had some extraordinary stories to tell which dated back decades, and it quickly became clear to me that some of my own annoyances in the past needed to be put into perspective a little more. On the call there were others who had gone through the cross linking experiences, and they proved incredibly helpful to get in contact with both before and after my procedure.
Over 4 years on and I continue to join the Zoom calls and go to the in-person meets when I can. Because of the group, I have learnt about many other types of procedures/contact lenses/eyes drops/helpful practices etc which I never knew existed. Thanks to the group, I learnt about mini scleral lenses. At one of my contact lens appointments, I pushed for me to try these out. I have now been wearing mini scleral lenses for over a year, and the vision and comfort that they have given me so far has been fantastic.
At these meetings/on these Zoom calls, I hear of worried parents whose children are struggling and they don’t want to know about their condition. I find it quite humorous as the stories often parallel my own experiences, but I also will these young adults to step forward, talk about their condition and learn from others. Perhaps they also need their own eureka moment like I did when it came to protecting my vision for driving, or perhaps they can read something like my story and see that there is help out there if you ask for it.
Volunteers Required for Research
What is this about?
We are conducting research to make immersive technologies more accessible for people with visual impairments. Insights about your daily challenges and your adaptive strategies are incredibly valuable for developing tools that would help to make such technology more accessible and inclusive.How can you help?
- Share your personal experience of living with a visual impairment: how you navigate various spaces, which challenges you encounter, and what strategies you employ to mitigate these challenges.
- Help us understand what makes technology truly accessible for you.
What to expect?
- One-hour online interview on Teams / Zoom or other platform of your choice. Please note that the interview would be recorded.
- No technical knowledge is required, we want to learn from your day-to-day experiences of living with a visual impairment.
- Follow-up questions and further discussions are welcome!
Why take part?While there is no monetary reward for taking part in this research, your contribution would influence the future of immersive technologies, making them more inclusive and accessible to visually impaired people.Interested in participating?Please contact Yuliya Chystaya at: yuliya.chystaya@brunel.ac.uk PhD candidate at Brunel University of London.
AGM and Speaker Meeting
Our next London meeting will be held on Saturday 22nd March at 11 am
The venue is at the Moorfields Education Hub, 1st Floor,
15 Ebenezer Street, N1 7NG (the hub is opposite Moorfields main hospital — cross City Road and go up Provost Street to the next corner and the Hub is facing you on the left).
Our guest speakers are
Dr Ahmed Abass and Lynn White.
They will be updating us on their project to make special spectacles for those of us looking for temporary relief from contact lenses. Their talk is titled
Non-orthogonal spectacle correction for irregular astigmatism
You can read more –here.
All are welcome, both people with KC and family members. As usual, there will be a buffet lunch and an opportunity to meet others with KC.
Do let us know if you are coming by emailing anne@kcgroup.org.uk or ‘phoning her on 020-8993 4759 so we can cater for numbers. If you are unsure whether the meeting is for you, please give Anne a call.
Note to Optometrists and ECLO’s – Please help us publicise this event by downloading and displaying a copy of this poster
Formal documentation for the AGM are available here:
Agenda
Minutes of last AGM
Chairmans Report
Draft Accounts
KC Zoom Meeting
The Next Meeting – Wednesday 10th September 2025
Our drop-in Zoom chats provide a good opportunity for the newly diagnosed to meet seasoned travelers and discuss all things KC. Anyone interested in Keratoconus is free to join us.
Clinician Pauline Bradford will be attending. Pauline is a contact lens optician with a wide range of experience.
After this event the next scheduled zoom meeting will be:
Thursday 4th December at 7:00pm
To take part, simply send an email to david@kcgroup.org.uk to request a link.
Please join us to discuss all things KC. We are totally open with no pre planned agenda. Ours is a truly bottom up organisation run by members, all of whom have KC or a close connection. There are no silly questions. If you are thinking it, then there is sure to be someone else with a similar idea.
We hope to see you then.
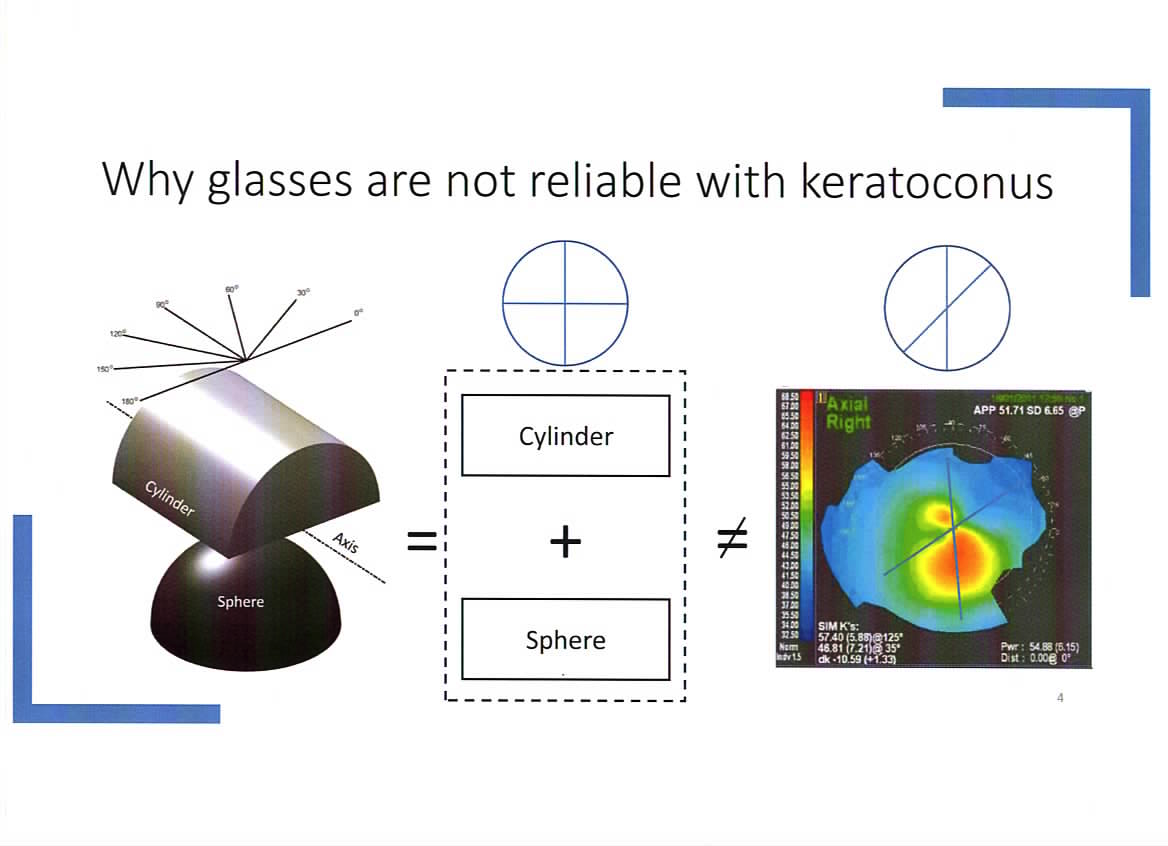
Spectacles for KC Research update 2
If you have been following this topic, you would have seen our earlier posts here and here
We are pleased to report that Dr Ahmed Abass has published his paper which we are proud to have co-sponsored with Fight for Sight.
Dr Abass has kindly provided the following simplified abstract. The full paper can be read here. You can also view his other papers on his University Page here
Purpose
The study aimed to investigate whether non-orthogonal correction in spectacles would improve the visual acuity of people with keratoconus. Non-orthogonal correction is where the angle between the cylinder powers of a spectacle lens for astigmatism is less than or greater than 90 degrees. Typically, keratoconus patients have this type of astigmatism, which is why standard glasses do not work well.
Methods
The study involved 18 patients with keratoconus from an eye clinic in Liverpool. After evaluating their eye condition, 23 eyes were selected for testing, while others were excluded due to the eye having little or no keratoconus or severe dryness. Each eye was tested first with standard lenses, and then with non-orthogonal lenses. The lenses were tested with different angles until the best one was found for each person. Participants were asked to rate their vision, specifically how clear letters appeared and how much “ghosting” (double vision) they experienced with each lens type.
Results
Of the eyes tested, 61% showed improved vision with non-orthogonal lenses, 30% saw no change, and 9% saw a slight decrease in vision. Regardless of vision improvement, 87% of participants noticed clearer letters, and 79% reported less ghosting. Most of the preferred angles for the non-orthogonal lenses were between 80° and 85°. The study also showed that for many people, the non-orthogonal lenses could provide a more accurate prescription compared to regular lenses. Nearly all participants said they would be interested in trying non-orthogonal glasses.
Conclusions
The study suggests that non-orthogonal lenses could improve vision and reduce issues like ghosting for people with keratoconus. While this is a promising step toward developing better glasses for these patients, more work is needed before these lenses can be made for everyday use.
We are hoping to arrange for Dr Abass to speak at one of our future events.
World Keratoconus Day
World Keratoconus Day is fast approaching on 10th November. We would love to hear from YOU! We are keen to support NKCF who this year are inviting everyone touched by keratoconus(KC) to join their 2024 World KC Day Contest – whether you are living with the condition, a supportive family member, friend, or an eye care professional who treats or studies KC. Share your unique story or perspective to help raise awareness and connect with others in the keratoconus community.
We encourage you to express your experience in a creative way.
| Here are some options to get started: |
How you can share:
Create a short Video
Write a poem
Design some digital artwork
Compose an essay
Or use any other medium that feels right for you
Ideas for what to share
Describe how keratoconus has impacted your life – both the challenges and the victories. A heartfelt thank you to someone who has supported you along the way. Giving a shout out to an eye care provider who has made a difference. Sharing your personal KC journey
Eye care professionals:
Why are you passionate about treating those with keratoconus?
Feel free to share anything else relating to your KC experience.
Your submission will be reviewed by representatives from NKFC, Keratoconus Barbados, Keratoconus Australia and the UK Keratoconus Group
Winners will be announced shortly after World Keratoconus day on 10th November 2024.
We can not wait to hear your stories and celebrate your creativity!
| To learn more or submit your entry, click below: World KC Contest. |